Electro-Federation Canada (EFC) is the voice for advocacy and the advancement of safe electrical products in Canada. EFC collaborates with government, certification agencies and regulators to promote the availability and safe use of approved electrical products. This portal serves to provide consumers, installers, regulators and other Canadians with timely resources and guidance on Germicidal UV (GUV) devices.
As requirements for clean, hygienic spaces become more critical to our health and wellbeing, leading manufacturers have developed GUV devices to help prevent the spread of infectious bacteria and viruses. There is an urgent need for GUV devices to be registered and certified before they can be sold in the Canadian market.
EFC members abide by safety and compliance regulations and support Health Canada’s commitment to protecting the safety of Canadians. As GUV devices become registered by Health Canada, this portal will provide quick links to manufacturers’ approved products.
GUV Devices for Advanced Health & Wellbeing
GUV devices provide a proven and effective means of disinfecting public and shared spaces. These devices revive the use of ultraviolet (UV) wavelengths to inactivate and prevent microorganisms from multiplying, thereby reducing the spread of harmful viruses and bacteria.

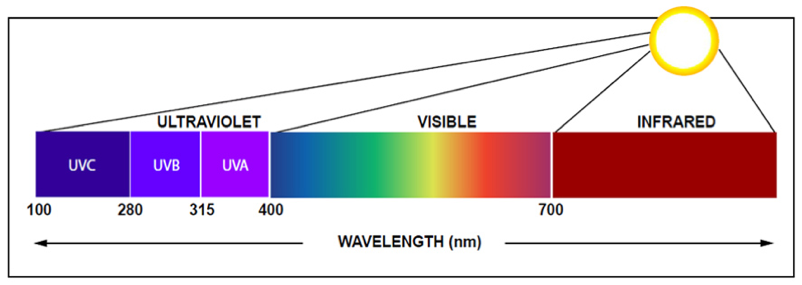
UV is transmitted at different wavelengths and frequencies: the short-wavelength (between 100 nm and 280 nm) is known as UV-C, which has been used in hospitals for decades to prevent drug-resistant superbugs from spreading and to disinfect surgical suites. UV-C has also been used in heating, cooling and ventilation systems to purify recirculated air. There is now a strong need to expand the use of UV-C by incorporating it into GUV devices that can sanitize a broader range of spaces to support our safe use of offices, schools, restaurants, stores and other shared spaces. View FAQs for more details
Become a Safety Ambassador
Do not assume that any marketed GUV device is registered or certified for sale, installation or use in Canada. Follow these two steps before purchasing or installing a GUV device:
- Check to see if it has been approved and registered by searching Health Canada’s product information database
- If you see a product that is not registered by Health Canada, report the product to Health Canada
Exemption: Health Canada requires registration of final GUV products. UV-C lamps and components are considered parts of GUV products, and therefore, do not need to be registered.
It Takes a Community to Champion Safety
EFC’s members are experts in designing, producing and selling products that exceed safety regulations and support Canada’s systematic testing and certification processes to safeguard consumers. EFC’s electrical distributors and sales agents work closely with manufacturer partners and customers to bring safe products to market. Explore EFC’s community of manufacturers, distributors and agents who are dedicated to advancing safety in the Canadian market.
FAQ: GUV Technology in Canada
This resource guide provides answers to frequently asked questions about GUV technology. If you have any further questions, please contact info@electrofed.com.